What is a Plasma?
Grade 6 Science Worksheets
Plasma is the fourth state of matter. A collection of positively charged ions and free electrons are produced in the high-energy state of matter known as plasma when electrons are stripped from their parent atoms.
What is a Plasma? - Grade 6 Science Worksheet PDF
This is a free printable / downloadable PDF worksheet with practice problems and answers. You can also work on it online.
|
Untimed | |
Sign up with your email ID to access this free worksheet.
"We really love eTutorWorld!"
"We really love etutorworld!. Anand S and Pooja are excellent math teachers and are quick to respond with requests to tutor on any math topic!" - Kieran Y (via TrustSpot.io)
"My daughter gets distracted easily"
"My daughter gets distracted very easily and Ms. Medini and other teachers were patient with her and redirected her back to the courses.
With the help of Etutorworld, my daughter has been now selected in the Gifted and Talented Program for the school district"
- Nivea Sharma (via TrustSpot.io)
Unlike solids, liquids, and gases, plasmas are determined by the behaviour of their charged particles rather than by their shape or volume. Plasmas are found frequently in stars, lightning, and fluorescent lights. They are also used in a wide range of commercial and technological processes, such as plasma displays and plasma cutting.
Discovery of Plasma
In the late 19th century, British physicist Sir William Crookes made the first discovery of plasma. When he noticed that cathode rays—streams of electrons generated in a vacuum tube—were making a gas glow, he started looking into them.
The Greek word for “moldable substance” inspired him to give this new state of matter the name “plasma.”
The study of plasmas and the creation of techniques for its containment and management by scientists and engineers over the ensuing decades produced a wide range of useful applications in fields like fusion energy, plasma cutting, and plasma displays.
How is Plasma different from other States of Matter
There are several ways in which plasma differs from solids, liquids, and gases:
- Contrary to solids, liquids, and gases, plasmas are made up of a mixture of positively charged ions and free electrons rather than neutral atoms or molecules.
- Shape and volume: Unlike solids, liquids, and gases, plasmas are not characterized by their shape or volume. Plasmas don’t have a set shape, and the behaviour of their charged particles determines how big they are.
- Energy: Plasmas are extremely energetic states of matter, capable of reaching millions of degrees Celsius in temperature. As a result of the electrons’ ability to separate from their parent atoms at this high energy level, ions and free electrons are produced.
- Due to the presence of free electrons that can move and carry an electrical charge, plasmas are excellent electrical conductors.
- Plasmas have a distinctive magnetic field response, which makes them useful in a variety of technological applications like plasma cutting and plasma displays.
These distinctions make plasma a special and extremely useful state of matter, distinguishing it from solids, liquids, and gases.
Physical Properties of Plasma
Plasmas’ physical characteristics include:
1. Due to the presence of free electrons, plasmas are excellent conductors of electricity.
2. Magnetic properties: Magnetic fields can affect plasmas, and plasmas can also produce their own magnetic fields.
3. Temperature: The high temperatures of plasmas, which are frequently measured in the millions of degrees Celsius, enable the electrons to separate from their parent atoms.
4. Ionization: Plasmas have special electrical and magnetic properties because they are made up of a combination of positively charged ions and free electrons.
5. Light Emission: Free electrons in plasmas collide with ions to excite them and produce photons, which are then emitted as light.
6. Plasma Oscillations: Collective oscillations, or waves of synchronized charged particles, can be seen in plasmas.
Plasmas are useful in a number of applications, such as fusion energy research, plasma cutting, and plasma displays, thanks to their physical characteristics.
Chemical Properties of Plasma
In contrast to solids, liquids, and gases, plasmas are high-temperature states of matter in which electrons have been severed from their parent atoms. As a result, plasmas lack the distinct chemical characteristics of these states.
However, due to the presence of free electrons and ions, chemical reactions in plasmas are often distinct from those in solids, liquids, and gases.
For instance, plasmas have the ability to ionize gas molecules, causing them to interact with the plasma’s other species. As a result, fresh chemical compounds may form and light may be produced.
Plasmas can also promote chemical reactions through a variety of processes, including heating, UV radiation, and electrical discharge.
A wide variety of processes, including plasma processing, material synthesis, and environmental remediation, use plasma-based chemical reactions.
Kinesthetic Properties of Plasma
Being an extremely hot, ionized state of matter, plasmas have special kinesthetic characteristics that set them apart from solids, liquids, and gases. Key kinesthetic characteristics of plasmas include:
1. Plasmas are extremely sensitive to electromagnetic fields and are capable of producing their own magnetic fields.
2. Plasmas are excellent electrical conductors, allowing electrical charges to move freely within the plasma.
3. Plasmas are capable of interacting with and emitting electromagnetic radiation, such as X-rays, ultraviolet light, and visible light.
4. Due to the presence of free electrons and ions, which can collide and produce fluid-like patterns, plasmas can display turbulence and fluid-like behaviour.
5. Instabilities: Different kinds of instabilities, like filamentation, can affect the behaviour and stability of plasmas.
6. Applications for these kinesthetic plasma characteristics are numerous and include plasma processing, material synthesis, fusion energy, and space physics.
“There have been times when we booked them last minute, but the teachers have been extremely well-prepared and the help desk at etutorworld is very prompt.
Our kid is doing much better with a higher score.”
6th Grade Tutoring
eTutorWorld offers Personalized Online Tutoring for Math, Science, English, and Standardised Tests.
Our Tutoring Packs start at just under $22.49 per hour, and come with a moneyback guarantee.
Schedule a FREE Trial Session, and experience quality tutoring for yourself. (No credit card required.)
Molecular Forces of Attraction in Plasma
1. Ions and unbound electrons define the peculiar state of matter known as plasma.
2. Plasma’s gravitational attraction is distinct from that of solids, liquids, and gases.
3. The electromagnetic force, which is produced by charged particles (ions and electrons) interacting with electromagnetic fields and one another, is the main force of attraction in plasma. Depending on the relative charges of the involved particles, this force can be both attractive and repellent.
4. Ions and electrons in plasmas are very mobile and can move around freely in response to magnetic and electric fields. In contrast to other states of matter, this results in the formation of complex structures and phenomena like electric currents, magnetic fields, and turbulence.
Due to these characteristics, plasma is a special and fascinating state of matter with a wide range of useful applications, such as fusion reactions, electric discharge lamps, plasma displays, and plasma cutting and welding.
Examples of Plasma
1. The Sun and other stars: The corona, the sun’s outer layer, is made of plasma and is where light and heat are emitted from the sun.
2. Lightning: Plasma is a type of electric discharge that occurs in lightning.
3. The ionosphere of the Earth: The aurora is formed by plasma, which is a component of the upper atmosphere of the Earth.
4. Plasma display devices: Plasma technology is used to create images on plasma displays, including televisions and computer monitors.
5. Plasma processing: Plasmas are used to change the characteristics of materials in a number of industrial processes, including plasma etching and plasma cutting.
6. Fusion energy: High-energy reactions between plasma particles in fusion reactors, which use plasma as the fuel, produce heat and electricity.
7. Plasma lamps: Plasma lamps produce light by creating a bright, glowing plasma out of a combination of gases.
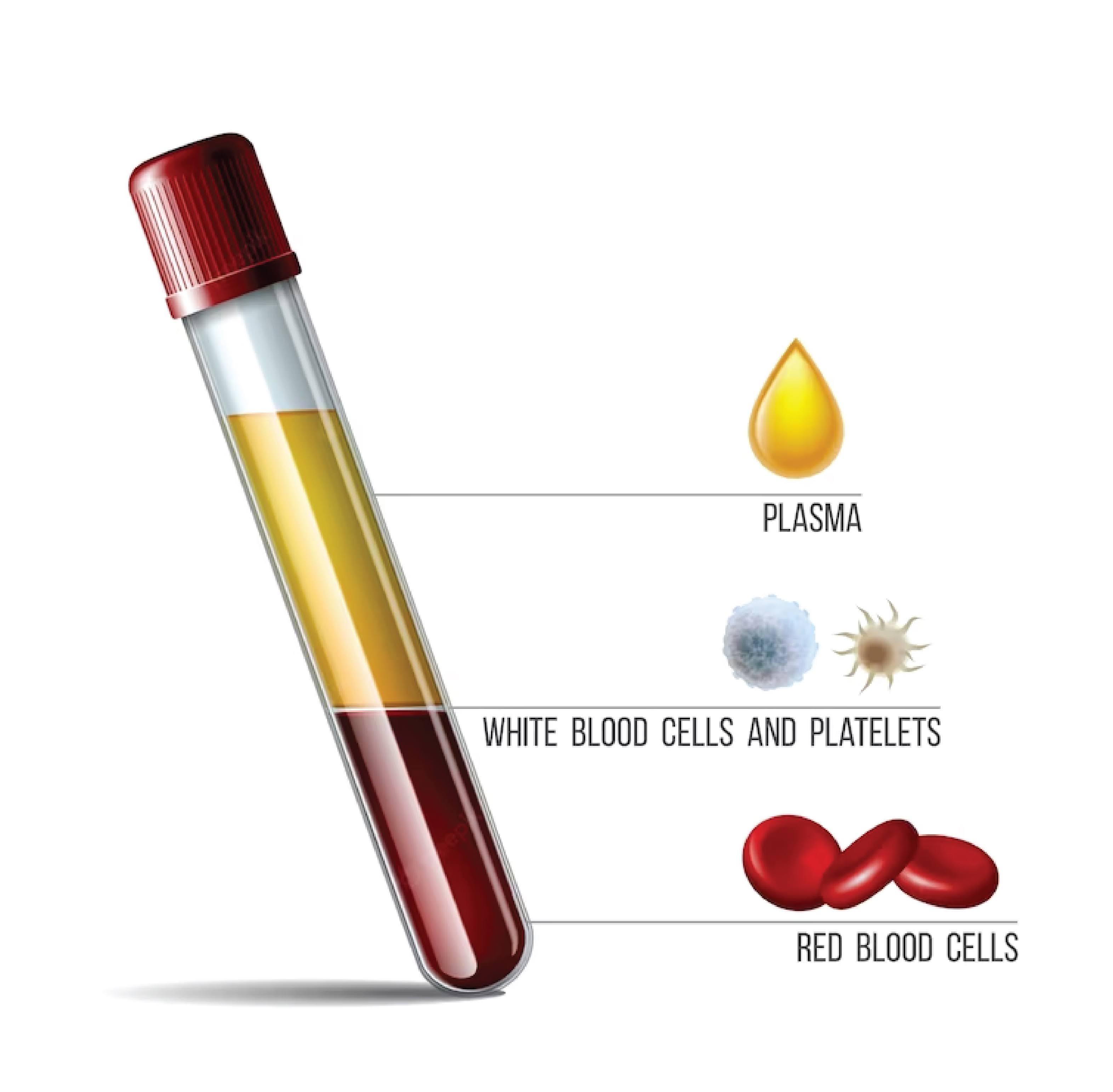
Is Blood Plasma?
Yes, a type of plasma is blood plasma. About 55% of the volume of blood is made up of blood plasma, which is a liquid substance. It is a yellowish liquid made up of proteins, salts, sugars, and water. Throughout the body, plasma serves as a transport medium for a variety of substances, including hormones, nutrients, waste products, and cells. Additionally, it helps to control blood pressure and maintains the body’s fluid and electrolyte balance.
Cool Experiments to perform with Plasma
The properties of plasma can be studied through a variety of experiments. Here are a few illustrations:
1. Glow discharge: To create a glow discharge in this experiment, a gas is added to a sealed chamber and a high voltage is applied. The gas is ionized by this discharge, producing a plasma that can be seen.
2. A plasma ball is a device that generates glowing plasma inside of a glass sphere using high voltage. You can see how the plasma responds to electric fields by touching the sphere.
3. In this experiment, two electrodes are used to spark one another, ionizing the air and generating plasma. The spark discharge can be used to study plasma characteristics, such as charged particle behaviour and the emergence of plasma instabilities.
4. High-power lasers are used in this experiment to produce plasma jets, which can be used to study the behaviour of plasma under challenging circumstances like high temperatures and pressures.
5. Insights gained from these experiments into the characteristics and behaviour of plasma are crucial for the creation of new technologies and applications based on plasma science.
Due to its special characteristics, plasma, the fourth state of matter, differs from solids, liquids, and gases. Plasma can be studied using a variety of experiments, such as glow discharges, plasma balls, spark discharges, and plasma jets.
These studies shed light on how charged particles behave in plasma and how plasma instabilities develop. In fields like plasma physics and plasma chemistry, the study of plasma is critical for the creation of new technologies and applications.
Do You Stack Up Against the Best?
If you have 30 minutes, try our free diagnostics test and assess your skills.
FAQS
What is plasma?
The presence of charged particles and ions in a gas-like environment defines plasma, the fourth state of matter. When a gas is heated to a high temperature, the electrons are stripped from the atoms, resulting in a partially ionized mixture.
Which physical characteristics does plasma possess?
The ability to emit light, electrical conductivity, and magnetic properties are just a few of the unusual physical characteristics of plasma. It is also highly thermally conductive, making it simple to transfer heat.
What are the plasma's chemical characteristics?
Because it is made up of ions and free electrons, plasma differs from solids, liquids, and gases in terms of its chemical characteristics. This makes plasma capable of chemical reactions that these other states of matter are not capable of.
What are some natural instances of plasma?
Lightning, the Sun, and the stars are a few typical examples of plasma in nature. Plasma in the ionosphere of Earth, comet tails, and the cores of gas giant planets are additional examples.
Can plasma be used in real-world applications?
Yes, a wide range of technologies and applications, such as electric arc welding, plasma cutting, plasma displays, and plasma spray coating, use plasma. Additionally, it is employed in the investigation of fusion reactors and in medical applications, such as blood plasma.

Kathleen Currence is one of the founders of eTutorWorld. Previously a middle school principal in Kansas City School District, she has an MA in Education from the University of Dayton, Ohio. She is a prolific writer, and likes to explain Science topics in student-friendly language. LinkedIn Profile
Affordable Tutoring Now Starts at Just $22.49
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12. We are also a leading provider of Test Prep help for Standardized Tests (SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE, and AP).
What makes eTutorWorld stand apart are: flexibility in lesson scheduling, quality of hand-picked tutors, assignment of tutors based on academic counseling and diagnostic tests of each student, and our 100% money-back guarantee.
Whether you have never tried personalized online tutoring before or are looking for better tutors and flexibility at an affordable price point, schedule a FREE TRIAL Session with us today.
*There is no purchase obligation or credit card requirement
Grade 6 Science Worksheets
- Inquiry process
- Nature of Science
- Scientific Inquiry
- Inquiry, Analysis and Problem Solving
- Ethical Practices
- Science and Society
- Biotic and Abiotic Factors
- Impact of Organisms
- Adaptation
- Spheres of Earth
- Natural Resources
- Environmental Issues
- Conservation of Earth
- Understanding Technology
- Abilities To Do Technological Design
- Structure of Earth
- Solar System
- Rocks and Fossils
- Earth Systems
- Plate Tectonics
- Evolution
- Magnetic Field of Earth
- Geologic Time
- Materials and Processes That Shape a Planet
- Astronomy
- Ecology
- Energy
- Kinetic and Potential Energy
- Energy Transfer
- Matter and its Structure
- States of Matter
- Physical and Chemical Changes
- Force and Motion
- Electricity and Magnetism
- Wave Interactions
- Sound
- Light
- Introduction to Life Science
- The Origin & History of Life On Earth
- Plant and Animal Cells
- Parts of a Cell
- The Cell Cycle
- How Living Organisms Get Energy
- Classification of Organisms
- How Plants Grow & Reproduce
- The Human Respiratory System
- The Human Cardiovascular System
- The Human Digestive System
- The Human Endocrine Systems
- The Human Nervous System
- The Human Muscular System
- The Human Skeletal System
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird










