Intensive Properties of Matter
Grade 6 Science Worksheets
Intensive properties are characteristics of matter that do not depend on the amount of matter present. These properties are the same for a given substance regardless of the size or amount of sample.
Intensive Properties of Matter - Grade 6 Science Worksheet PDF
This is a free printable / downloadable PDF worksheet with practice problems and answers. You can also work on it online.
|
Untimed | |
Sign up with your email ID to access this free worksheet.
"We really love eTutorWorld!"
"We really love etutorworld!. Anand S and Pooja are excellent math teachers and are quick to respond with requests to tutor on any math topic!" - Kieran Y (via TrustSpot.io)
"My daughter gets distracted easily"
"My daughter gets distracted very easily and Ms. Medini and other teachers were patient with her and redirected her back to the courses.
With the help of Etutorworld, my daughter has been now selected in the Gifted and Talented Program for the school district"
- Nivea Sharma (via TrustSpot.io)
Before we delve into understanding the intensive properties of matter. Let us revise the matter.
Define matter
The matter is anything that takes up space and has mass. It can be found all around us and comes in many different forms. Matter can be solid, like a rock or pencil; liquid, like water or juice; and gas, like air or steam. Even things we can’t see, like the air we breathe, are made of matter.
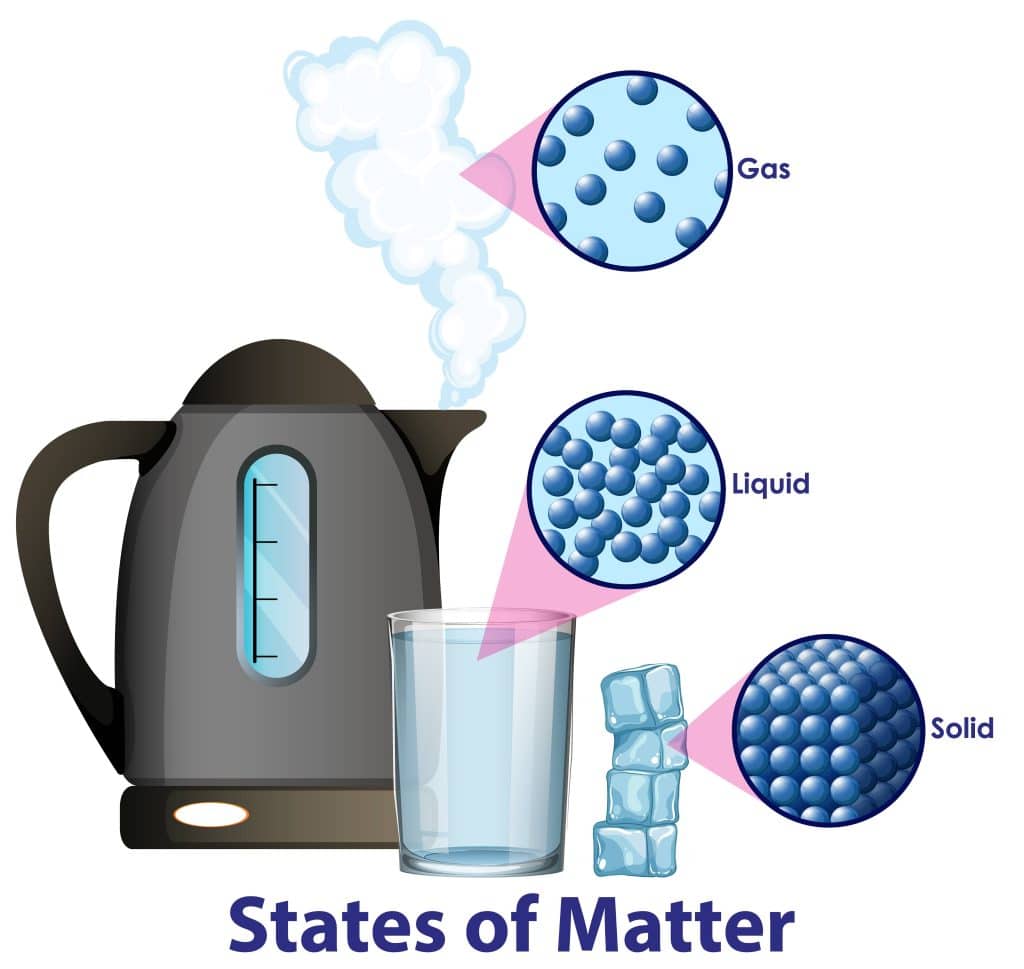
Examples of matter are the table you are sitting on, the chair you are sitting on, the pencil and paper you are using, the water you drink, the air you breathe, and so on. Everything you can touch, see, smell or taste is made of matter.
What is the Intensive Property of Matter?
Intensive properties are characteristics of matter that do not depend on the amount of matter present. These properties are the same for a given substance regardless of the size or amount of sample.
Examples of some intensive properties include:
Density: The ratio of mass to volume, which is the same for a given substance regardless of the amount of matter present. For example, the density of gold is 19.3 g/cm3, whether we have a small piece or a large bar of gold.

Melting point: The temperature at which a solid becomes a liquid. This property is the same for a given substance regardless of the amount of matter present. For example, the melting point of ice is 0 °C, whether we have a small cube or a large block of ice.
“There have been times when we booked them last minute, but the teachers have been extremely well-prepared and the help desk at etutorworld is very prompt.
Our kid is doing much better with a higher score.”
6th Grade Tutoring
eTutorWorld offers Personalized Online Tutoring for Math, Science, English, and Standardised Tests.
Our Tutoring Packs start at just under $22.49 per hour, and come with a moneyback guarantee.
Schedule a FREE Trial Session, and experience quality tutoring for yourself. (No credit card required.)
Boiling point: The temperature at which a liquid changes to gas. This property is the same for a given substance regardless of the amount of matter present. For example, the boiling point of water is 100 °C, whether we have a small droplet or a large volume of water.
Hardness: The resistance of a material to being scratched or indented. This property is the same for a given substance regardless of the amount of matter present.
For example, the hardness of a diamond is 10 (on the Mohs scale) whether we have a small grain or a large diamond.
In summary, intensive properties are matter characteristics independent of the amount of matter present. They are the same for a given substance regardless of the size or amount of sample.
Do You Stack Up Against the Best?
If you have 30 minutes, try our free diagnostics test and assess your skills.
Intensive Properties of Matter FAQS
What are the intensive properties of matter?
Intensive properties are characteristics of matter that do not depend on the amount of matter present. They are the same for a given substance regardless of the size or amount of sample. Examples include density, melting point, boiling point, and hardness.
How are intensive properties different from extensive properties?
Intensive properties are independent of the amount of matter present, while extensive properties depend on the amount of matter present.
Can intensive properties be used to identify a substance?
Yes, intensive properties can be used to identify a substance because they are unique to that substance and do not change with the amount of matter present.
Do intensive properties change with temperature or pressure?
Some intensive properties may change with temperature or pressure; for example, density of a gas changes with temperature and pressure.
How can intensive properties be measured?
Intensive properties can be measured using various techniques such as density measurement, calorimetry, and hardness testing.

Kathleen Currence is one of the founders of eTutorWorld. Previously a middle school principal in Kansas City School District, she has an MA in Education from the University of Dayton, Ohio. She is a prolific writer, and likes to explain Science topics in student-friendly language. LinkedIn Profile
Affordable Tutoring Now Starts at Just $22.49
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12. We are also a leading provider of Test Prep help for Standardized Tests (SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE, and AP).
What makes eTutorWorld stand apart are: flexibility in lesson scheduling, quality of hand-picked tutors, assignment of tutors based on academic counseling and diagnostic tests of each student, and our 100% money-back guarantee.
Whether you have never tried personalized online tutoring before or are looking for better tutors and flexibility at an affordable price point, schedule a FREE TRIAL Session with us today.
*There is no purchase obligation or credit card requirement
Grade 6 Science Worksheets
- Inquiry process
- Nature of Science
- Scientific Inquiry
- Inquiry, Analysis and Problem Solving
- Ethical Practices
- Science and Society
- Biotic and Abiotic Factors
- Impact of Organisms
- Adaptation
- Spheres of Earth
- Natural Resources
- Environmental Issues
- Conservation of Earth
- Understanding Technology
- Abilities To Do Technological Design
- Structure of Earth
- Solar System
- Rocks and Fossils
- Earth Systems
- Plate Tectonics
- Evolution
- Magnetic Field of Earth
- Geologic Time
- Materials and Processes That Shape a Planet
- Astronomy
- Ecology
- Energy
- Kinetic and Potential Energy
- Energy Transfer
- Matter and its Structure
- States of Matter
- Physical and Chemical Changes
- Force and Motion
- Electricity and Magnetism
- Wave Interactions
- Sound
- Light
- Introduction to Life Science
- The Origin & History of Life On Earth
- Plant and Animal Cells
- Parts of a Cell
- The Cell Cycle
- How Living Organisms Get Energy
- Classification of Organisms
- How Plants Grow & Reproduce
- The Human Respiratory System
- The Human Cardiovascular System
- The Human Digestive System
- The Human Endocrine Systems
- The Human Nervous System
- The Human Muscular System
- The Human Skeletal System
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird










