Law of Conservation of Mass
Grade 7 Science Worksheets
The Law of Conservation of Mass states that the total mass in a closed system remains constant.
Table of Contents:
- Law of Conservation of Mass
- Formula for Law of Conservation of Mass
- FAQs
Law of Conservation of Mass - Grade 7 Science Worksheet PDF
This is a free printable / downloadable PDF worksheet with practice problems and answers. You can also work on it online.
|
Untimed | |
Sign up with your email ID to access this free worksheet.
"We really love eTutorWorld!"
"We really love etutorworld!. Anand S and Pooja are excellent math teachers and are quick to respond with requests to tutor on any math topic!" - Kieran Y (via TrustSpot.io)
"My daughter gets distracted easily"
"My daughter gets distracted very easily and Ms. Medini and other teachers were patient with her and redirected her back to the courses.
With the help of Etutorworld, my daughter has been now selected in the Gifted and Talented Program for the school district"
- Nivea Sharma (via TrustSpot.io)
Law of Conservation of Mass
The law of conservation of mass means that the mass of the products of a chemical reaction or physical process will always be equal to the mass of the reactants.
For example, if you burn a piece of wood, the mass of the ash, smoke, and gases produced will be equal to the mass of the wood that was burned. Even though the wood is destroyed, the mass is still there, just in a different form.
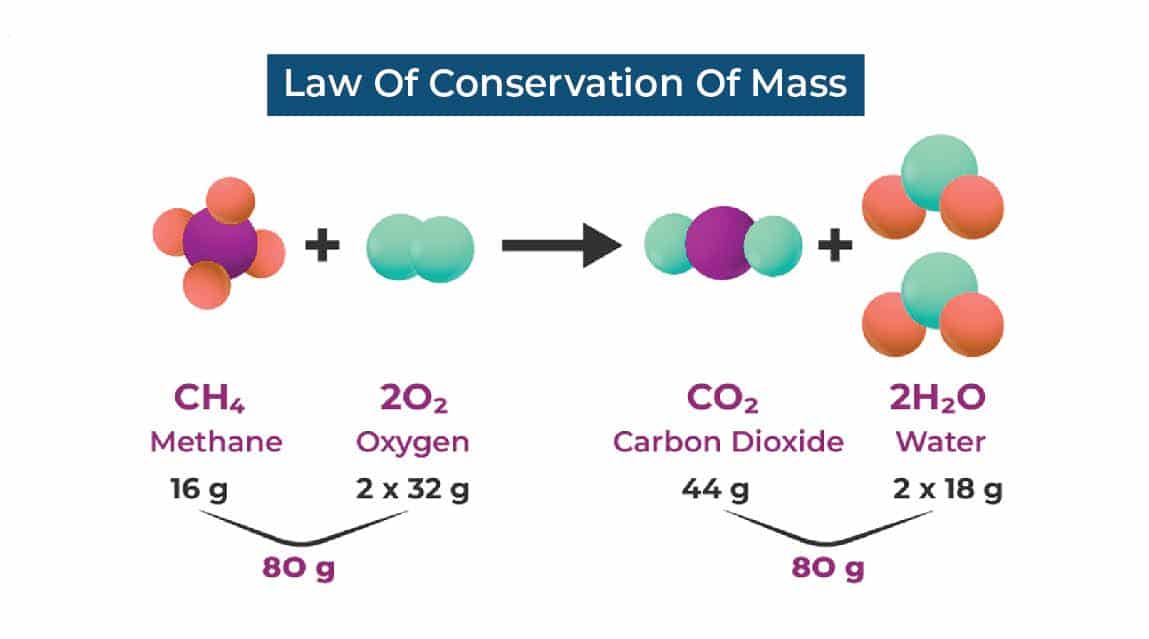
Another example is when a mixture of hydrogen and oxygen gases are combined to make water, the mass of the water formed will be equal to the total mass of the hydrogen and oxygen gases.
In simple terms, the law of conservation of mass states that the total amount of mass in a closed system remains constant and it can be converted from one form to another but cannot be created or destroyed.
“There have been times when we booked them last minute, but the teachers have been extremely well-prepared and the help desk at etutorworld is very prompt.
Our kid is doing much better with a higher score.”
7th Grade Tutoring
eTutorWorld offers Personalized Online Tutoring for Math, Science, English, and Standardised Tests.
Our Tutoring Packs start at just under $21 per hour, and come with a moneyback guarantee.
Schedule a FREE Trial Session, and experience quality tutoring for yourself. (No credit card required.)
Formula for Law of Conservation of Mass
The formula for the law of conservation of mass is:
mass of reactants = mass of products
This formula can be expressed mathematically as:
m_reactants = m_products
Where m represents the mass of the reactants or products.
For example, if you have 100 grams of hydrogen gas (H2) and 200 grams of oxygen gas (O2) that react to form water (H2O), the mass of the reactants and products can be calculated as follows:
mass of reactants (H2 + O2) = 100 grams + 200 grams = 300 grams
mass of products (H2O) = 300 grams
As you can see, the mass of the reactants (hydrogen and oxygen gas) is equal to the mass of the product (water), which confirms that the law of conservation of mass holds true.
Another example, if you have 20 grams of magnesium metal (Mg) and 40 grams of oxygen gas (O2) that react to form magnesium oxide (MgO), we can calculate the mass of the reactants and products as follows:
mass of reactants (Mg + O2) = 20 grams + 40 grams = 60 grams
mass of products (MgO) = 60 grams
As you can see, the mass of the reactants (magnesium metal and oxygen gas) is equal to the mass of the product (magnesium oxide), which confirms that the law of conservation of mass holds true.
Do You Stack Up Against the Best?
If you have 30 minutes, try our free diagnostics test and assess your skills.
Law of Conservation of Mass FAQS
What is the law of conservation of mass?
The law of conservation of mass states that the total mass in a closed system remains constant, meaning that the mass of the products of a chemical reaction or physical process will always be equal to the mass of the reactants.
How is the law of conservation of mass related to the law of conservation of energy?
The law of conservation of mass is related to the law of conservation of energy in that both principles state that certain quantities (mass and energy, respectively) cannot be created or destroyed, only transferred or converted from one form to another.
Can mass be lost in a system?
Mass cannot be lost in a closed system, but it can be converted from one form to another. For example, in a chemical reaction, the mass of the reactants will be equal to the mass of the products.
What are some examples of mass conservation in everyday life?
Examples of mass conservation in everyday life include baking a cake (the mass of the ingredients is equal to the mass of the final product), burning wood in a fireplace (the mass of the wood is equal to the mass of the ash and gases produced), and the burning of fossil fuels (the mass of the fuel is equal to the mass of the ash and gases produced).
How does the law of conservation of mass apply to living things?
The law of conservation of mass applies to living things in the sense that the mass they consume must come from somewhere. For example, plants take in carbon dioxide and water, which they use to make carbohydrates and oxygen through photosynthesis. The mass of the carbohydrates and oxygen is equal to the mass of the carbon dioxide and water consumed.

Kathleen Currence is one of the founders of eTutorWorld. Previously a middle school principal in Kansas City School District, she has an MA in Education from the University of Dayton, Ohio. She is a prolific writer, and likes to explain Science topics in student-friendly language. LinkedIn Profile
Affordable Tutoring Now Starts at Just $22.49
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12. We are also a leading provider of Test Prep help for Standardized Tests (SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE, and AP).
What makes eTutorWorld stand apart are: flexibility in lesson scheduling, quality of hand-picked tutors, assignment of tutors based on academic counseling and diagnostic tests of each student, and our 100% money-back guarantee.
Whether you have never tried personalized online tutoring before or are looking for better tutors and flexibility at an affordable price point, schedule a FREE TRIAL Session with us today.
*There is no purchase obligation or credit card requirement
Grade 7 Science Worksheets
- Elements and Compounds
- Solar Energy
- Photosynthesis
- Electricity and Magnetism
- Law of conservation of energy
- Periodic table
- Properties of Matter
- Waves
- Energy Resources
- Weather and Climate
- Immune, Circulatory and Digestive Systems
- Organs in Multi-cellular Organism
- Sedimentary, Igneous, and Metamorphic Rocks
- Structure of the Earth
- Law of Conservation of Mass
- Physical and Chemical Changes
- Scientific Method
- Human Digestive System
- Environmental Science
- Renewable and Non-renewable energy Resources
- Characteristics of Living Organisms
- Life Science
- Earth and Space Science
- Solar Eclipse
- Heat Technology
- Newton’s Laws of Motions
- Physical Science
- Tools, Measurement and SI Units
- Earth Atmosphere
- Interactions of Living things
- The Earth Ecosystem
- Organelles in Plant and Animal cells
- Layers of the Earth
- Cycles in Nature
Grade 7 Math Worksheets
- Fractions
- Linear equations word problems
- Statistics
- Properties of Parallel Line
- Finding slope from an equation
- Identifying Quadrilaterals
- Percent Change
- Properties of addition and multiplication
- Pythagorean Theorem
- Solving two step inequalities
- Symmetry
- Fractions to Decimals (New)
- Whole Number Exponents with Integer Bases (New)
- Adding and Subtracting Fractions (New)
- Integer Addition and Subtraction (New)
- Dividing Mixed Numbers (New)
- Basics of Coordinate Geometry (New)
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird










