Changes in States of Matter
Grade 8 Science Worksheets
How does a Mercury Thermometer Work?
A thermometer shows temperature. The mercury inside the thermometer is sensitive to temperature – it expands when the temperature rises and contracts when it falls. This tells us how hot or cold it is around us.
The thermometer is an example of the effect of heat on matter.
Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
Effect of Heat on Matter
There are primarily three physical states of matter – solid, liquid and gas. The tiniest of particles of matter – atoms – combine to form molecules, which are the building blocks for all forms of matter. These atoms and molecules are in constant motion.
When the matter is heated, the atoms and molecules start to vibrate even faster. As a result of increased molecular motion, the space between them increases. The matter expands and occupies more space. The motion and spacing determine the state of the matter.
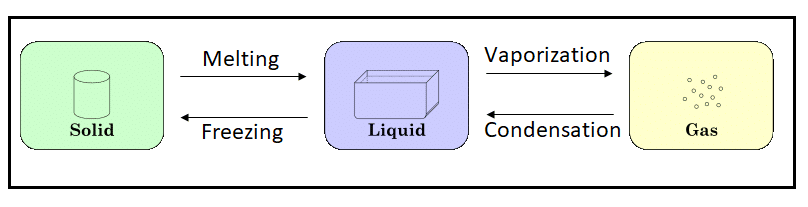

Continuous heating of solid matter will melt the substance to form a liquid. Gold, when heated, past its Melting Point, will form liquid gold. The chemical composition of the substance remains unchanged – gold in liquid form is still gold atoms and molecules, nothing else. Only the physical form changes.
Sustained heating of a liquid past its Boiling Point will vaporize the substance into gas.
Just as heating transforms the state of matter from solid to liquid to gas, so also, cooling transforms a gas to liquid to solid. A gas which is cooled to below its Condensation Point forms liquid. A liquid which is cooled to below its Freezing Point forms solid.
Learn more about Changes in States of Matter and other important topics with 8th Grade Science Tutoring at eTutorWorld. Our expert science tutors break down the topics through interactive one-to-one sessions. We also offer the advantage of customized lesson plans, flexible schedules and convenience of learning from home.
eTutorWorld Understands Math Tutoring | Online Math Worksheets are Important Tools
Understanding graphs, charts, and opinion polls in a newspaper, for calculating house and car payments, and for choosing a long-distance telephone service are impossible without strong math skills …and the only way to develop strong math skills is by constant practice.
‘Practice makes a man perfect’ holds true for no other field better than for math. A middle or high school student must set aside a minimum of an hour for math every day. Other than textbooks, worksheets help you revise and understand concepts better.
Our expert tutors prepare online maths worksheets that are age and grade-appropriate. Grade-wise math worksheets for Elementary Math, Arithmetic, Pre-Algebra, Algebra, Geometry, Trigonometry, Statistics, Pre-Calculus and Calculus can be solved to improve math skills, to get ahead or to even catch up.
You may download these FREE online math worksheets in the PDF format, and then print and email us their solutions for a free evaluation and analysis by eTutorworld’smath expert tutors.
You may solve these worksheets by yourself or with your peers while studying together.
The Answer Key at the end of each worksheet allows for a self-evaluation.
Personalized Online Tutoring
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12, Test Prep help for Standardized tests like SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE and AP. You may schedule online tutoring lessons at your personal scheduled times, all with a Money-Back Guarantee. The first one-on-one online tutoring lesson is always FREE, no purchase obligation, no credit card required.
For answers/solutions to any question or to learn concepts, take a FREE CLASS.
No credit card required, no obligation to purchase.
Just book a free class to meet a tutor and get help on any topic you want!

The Changing State of Water
Water exists in three states in nature. It is abundantly seen in its liquid form. In the liquid state it has a definite volume but can change its shape easily. The molecules of water are close to each other yet able to flow. It has a certain density. It cannot be compressed easily. And it is definitely visible.
Water has a boiling point of 100o Celsius (212oF). When water is heated to its boiling point you see it in the form of steam. The water from water bodies evaporates to form water vapor. Both are gaseous state of water. In this state, it has no definite volume or shape. It is also invisible. It can however be compressed easily.
Water has a freezing point of 0o Celsius (32oF). When water is cooled below its freezing point it forms ice, a solid. Water molecules are densely packed in its solid form. Although liquids contract when cooled, ice expands when it freezes because the molecules of water form a geometric pattern that takes up more space.
Check Point
- A solid which is heated past its melting point becomes ______.
- A gas which is cooled below its condensation point becomes ______.
- A liquid which is cooled below its freezing point becomes _______.
- A liquid which is heated above its boiling point becomes _______.
- The boiling point of water is _______ degrees Fahrenheit.
Answer Key
- Liquid
- Liquid
- Solid
- Gas
- 212
Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
Pricing for Online Tutoring
| Tutoring Package | Validity | Grade (1-12), College |
|---|---|---|
| 5 sessions | 1 Month | $139 |
| 1 session | 1 Month | $28 |
| 10 sessions | 3 months | $269 |
| 15 sessions | 3 months | $399 |
| 20 sessions | 4 months | $499 |
| 50 sessions | 6 months | $1189 |
| 100 sessions | 12 months | $2249 |
8th Grade Free Worksheets
- The Universe
- Heredity
- Evolutionary Theory
- Structure of the atom
- Ethical Practices
- Unveiling the mystery behind the physical universe
- Components of the universe
- Celestial phenomena
- The tilt of Earth’s axis
- The causes of high and low tides
- Earth Systems
- Rocks and Fossils
- Weather and Climate
- Basics of chemical reactions
- Types of Chemical reactions – Endothermic, exothermic, oxidation, reduction reactions
- Catalysts and enzymes
- Compounds and mixtures
- Acids, Bases and pH Indicators
Images Credit:
https://upload.wikimedia.org/wikipedia/commons/thumb/8/8f/Physics_matter_state_transition_1_en.svg/1064px-Physics_matter_state_transition_1_en.svg.png
https://www.nyu.edu
https://media.istockphoto.com/vectors/diagrame-of-matter-in-different-states-vector-id615287478
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird






