Acids, Bases and pH Indicators
Grade 8 Science Worksheets
Acids and Bases are groups of chemical compounds, which react with each other to form salts and water. The reaction is known as a Neutralization process – in other words, acids and bases neutralize one another.
Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
Acids
Acid solutions have a sour taste, they produce a burning sensation in contact with skin, and they are capable of dissolving many metals. Acids can be useful and also dangerous and poisonous.
Hydrochloric Acid(HCl) in the human stomach helps in the process of digestion. Vinegar is Acetic Acid (CH3COOH), which is used in salad dressings and for food preservation, and we obtain Citric Acid(C6H8O7) from oranges and lemons. Many types of cola contain Phosphoric Acid (H3PO4).
Sulfuric Acid (H2SO4), which is extremely harmful and corrosive to skin, is used in car batteries while Nitric Acid (HNO3) is used in explosives and fertilizers.
Acids are classified as Organic if they contain carbon atoms, else they are termed Inorganic. Organic acids are used in beverages, cosmetics, detergents, foods, drugs, soaps, and plastics. Inorganic acids are generally much stronger and are used to manufacture paints, explosives, fertilizers, and other chemicals.
Bases or Alkalis
Bases are substances that react with an acid to decrease or neutralize its acidic properties. Bases dissolved in water are called Alkalis or Alkaline Solutions. Bases have a slippery texture and taste bitter.
Bases are also very useful chemicals. Sodium Hydroxide [NaOH] is used in the manufacture of soap, paper and synthetic materials. Calcium hydroxide [Ca(OH)2] is used in bleaching powder and to neutralize soil acidity. Magnesium hydroxide [Mg(OH)2] is a common laxative.
Learn more about Acids, Bases and pH Indicators and other important topics with 8th Grade Science Tutoring at eTutorWorld. Our expert science tutors break down the topics through interactive one-to-one sessions. We also offer the advantage of customized lesson plans, flexible schedules and convenience of learning from home.
eTutorWorld Understands Math Tutoring | Online Math Worksheets are Important Tools
Understanding graphs, charts, and opinion polls in a newspaper, for calculating house and car payments, and for choosing a long-distance telephone service are impossible without strong math skills …and the only way to develop strong math skills is by constant practice.
‘Practice makes a man perfect’ holds true for no other field better than for math. A middle or high school student must set aside a minimum of an hour for math every day. Other than textbooks, worksheets help you revise and understand concepts better.
Our expert tutors prepare online maths worksheets that are age and grade-appropriate. Grade-wise math worksheets for Elementary Math, Arithmetic, Pre-Algebra, Algebra, Geometry, Trigonometry, Statistics, Pre-Calculus and Calculus can be solved to improve math skills, to get ahead or to even catch up.
You may download these FREE online math worksheets in the PDF format, and then print and email us their solutions for a free evaluation and analysis by eTutorworld’smath expert tutors.
You may solve these worksheets by yourself or with your peers while studying together.
The Answer Key at the end of each worksheet allows for a self-evaluation.
Personalized Online Tutoring
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12, Test Prep help for Standardized tests like SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE and AP. You may schedule online tutoring lessons at your personal scheduled times, all with a Money-Back Guarantee. The first one-on-one online tutoring lesson is always FREE, no purchase obligation, no credit card required.
For answers/solutions to any question or to learn concepts, take a FREE CLASS.
No credit card required, no obligation to purchase.
Just book a free class to meet a tutor and get help on any topic you want!
Acids and Bases in Chemistry
Acids dissolve in water to produce hydrogen ions. A hydrogen ion is a proton formed when a hydrogen atom loses its electron. Acid is also defined as a Proton Donor – it readily gives up a proton (hydrogen ion) to another substance. A base is able to accept a hydrogen ion from an acid – it is a Proton Acceptor.
However, all acids may not be able to release hydrogen and may not always be proton donors. In broader terms, therefore, all acids are defined as Electron-Pair Acceptors while bases are defined as Electron-Pair Donors – that is, acids can accept a pair of electrons from another atom or molecule while bases can share electron pairs with acids.
There are Strong Acids and Weak Acids, Strong Bases, and Weak Bases. Weak acids and bases do not ionize completely when dissolved in water, while strong acids are fully ionized. Sulfurous Acid (H2SO3) and Nitrous Acid(HNO2) are weak acids but sulfuric acid and nitric acid are strong. Weak acids like vinegar can be consumed without any problem but strong acids like hydrochloric acid will burn skin in touch.
The pH of Acids and Bases
pH, which stands for Potential(or Power) of Hydrogen, is a number that indicates the concentration of hydrogen ions in a solution. The concentration is expressed in moles of hydrogen ions per liter of solution.
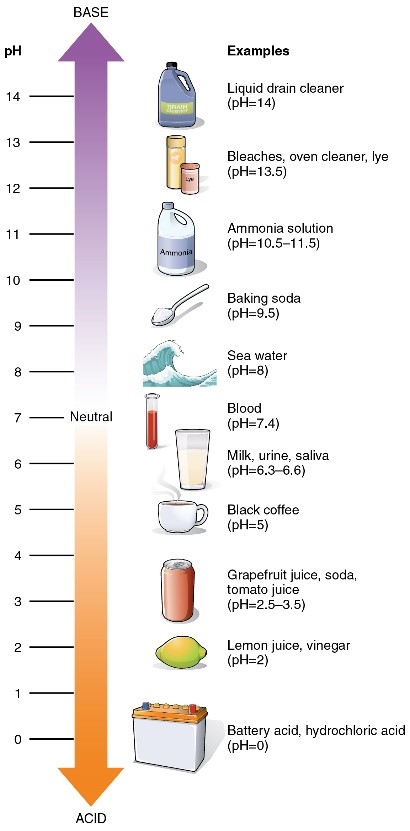
pH ranges from 0 to 14. A pH below 7 indicates that a solution is acidic while above 7 indicates that it’s basic (alkaline). A pH of 7 indicates a perfectly neutral solution – neither acidic nor basis – pure water, for example.
pH Indicators
Indicators are compounds that change color when they come in contact with acids and bases. They are therefore useful in detecting acids or acidity of solutions.
The most commonly used natural indicator is a dye named Litmus obtained from lichens – slow-growing plants on rocks, walls, and trees. Litmus has a mauve color in pure water. It turns red in an acidic solution and blue in alkaline solutions. Another natural indicator is Turmeric, a yellow-colored common kitchen spice, which remains yellow when dipped in acid but turns red in alkalis.
Many fruits and vegetables containing a group of pigments called Anthocyanins change color in response to pH. Red cabbage, grape juice, beet, tomato, and skins of peach, pear, plum, radish, and turnip are just a few. Pigments extracted from these can be made into juice or paper to conduct pH tests in homes and labs.
Chemical compounds like Phenolphthalein, Methyl Red, Methyl Orange, Thymol Blue, and many others also change color depending on whether they are added to acidic or basic solutions. They are frequently used in lab experiments.
Fun Fact: Hydrangea or snowball flowers are blue when grown in acidic soils and pink or red in basic soils.
Check Point
- Which is an organic acid?
- Acetic acid (CH3COOH)
- Phosphoric Acid (H3PO4)
- Nitric Acid (HNO3)
- Bases dissolved in water are called _______ solutions.
- Acids are Electron-Pair _______ while bases are defined as Electron-Pair _______.
- Which of these is alkaline?
- Grapefruit juice
- Ammonia solution
- Saliva
- Black coffee
- Litmus turns ______ in an acidic solution.
Answer Key
- a) Acetic acid (CH3COOH)
- Alkaline
- Acceptors, Donors
- b) Ammonia solution
- Red
Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
Pricing for Online Tutoring
| Tutoring Package | Validity | Grade (1-12), College |
|---|---|---|
| 5 sessions | 1 Month | $139 |
| 1 session | 1 Month | $28 |
| 10 sessions | 3 months | $269 |
| 15 sessions | 3 months | $399 |
| 20 sessions | 4 months | $499 |
| 50 sessions | 6 months | $1189 |
| 100 sessions | 12 months | $2249 |
8th Grade Free Worksheets
- The Universe
- Heredity
- Evolutionary Theory
- Structure of the atom
- Ethical Practices
- Unveiling the mystery behind the physical universe
- Components of the universe
- Celestial phenomena
- The tilt of Earth’s axis
- The causes of high and low tides
- Earth Systems
- Rocks and Fossils
- Weather and Climate
- Basics of chemical reactions
- Types of Chemical reactions – Endothermic, exothermic, oxidation, reduction reactions
- Catalysts and enzymes
- Compounds and mixtures
- Acids, Bases and pH Indicators
Images Credit:
https://upload.wikimedia.org/wikipedia/commons/2/23/216_pH_Scale-01.jpg
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird






