Catalysts and Enzymes
Grade 8 Science Worksheets
Speeding up Chemical Reactions
Ever noticed that sugar dissolves faster in hot water than in cold? Or that food cooks quicker on high flame? Temperature is an important factor in these matters. It is equally important in many chemical reactions. It has been experimentally observed that every 50oFrise in temperature doubles,and sometimes triples, the speed of chemical reactions.
Temperature is a measure of the average kinetic energy of molecules. At higher temperatures, molecules in motion tend to remain in motion, thereby increasing their ability to collide with molecules of other reactants to activate a chemical reaction. This is the Collision Theory, according to which molecules only react if they have sufficient energy for a reaction to take place.

Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
There are other ways to speed up chemical reactions –
- Increasing the concentration of reactants – more molecules are available for collision
- Increasing the surface area of reactants – larger surface area means greater collisions
Very often, chemists use an additional substance to alter the rate of chemical reactions. This additional substance is called a Catalyst and the chemical process using a catalyst is known as Catalysis.
Catalysts
Catalysts are used in industry to speed up many chemical reactions which otherwise would take place too slowly.
These are some of their inherent characteristics –
- A catalyst is purposeful in nature – a specific catalyst works only for a specific reaction.
- A catalyst provides a chemical pathway for a reaction to proceed far more rapidly in its presence than in its absence.
- Often, a catalyst reacts with one of the reactants to produce an intermediate chemical product which then reacts more readily with the other reactant to form the desired end product.
- A catalyst is not consumed by the chemical reaction, nor is it a part of the resulting products of the reaction in anyway.
Catalysts have a wide variety of uses in our daily lives and in industry –
- Cobalt-based catalysts to make smooth paper from wood pulp
- Zinc chloride as a catalyst to make synthetic fibers like nylon and rayon
- Catalysts in the manufacture of a wide variety of plastics from milk jugs to CDs to helmets
- Ammonia synthesis – nitrogen reacts with hydrogen at high temperature and pressure in the presence of iron as a catalyst to produce liquid ammonia
- Manufacture of Sulfuric acid – Sulfur dioxide and oxygen, passed over hot vanadium oxide as a catalyst, unite to form sulfur trioxide, which in turn combines with water to make sulfuric acid.
- Adding potassium permanganate as a catalyst to hydrogen peroxide hastens its decomposition into water and oxygen gas.
- Cars use catalytic converters containing platinum, which converts harmful carbon monoxide gas into less harmful carbon dioxide gas.
Learn more about Catalysts and Enzymes and other important topics with 8th Grade Science Tutoring at eTutorWorld. Our expert science tutors break down the topics through interactive one-to-one sessions. We also offer the advantage of customized lesson plans, flexible schedules and convenience of learning from home.
eTutorWorld Understands Math Tutoring | Online Math Worksheets are Important Tools
Understanding graphs, charts, and opinion polls in a newspaper, for calculating house and car payments, and for choosing a long-distance telephone service are impossible without strong math skills …and the only way to develop strong math skills is by constant practice.
‘Practice makes a man perfect’ holds true for no other field better than for math. A middle or high school student must set aside a minimum of an hour for math every day. Other than textbooks, worksheets help you revise and understand concepts better.
Our expert tutors prepare online maths worksheets that are age and grade-appropriate. Grade-wise math worksheets for Elementary Math, Arithmetic, Pre-Algebra, Algebra, Geometry, Trigonometry, Statistics, Pre-Calculus and Calculus can be solved to improve math skills, to get ahead or to even catch up.
You may download these FREE online math worksheets in the PDF format, and then print and email us their solutions for a free evaluation and analysis by eTutorworld’smath expert tutors.
You may solve these worksheets by yourself or with your peers while studying together.
The Answer Key at the end of each worksheet allows for a self-evaluation.
Personalized Online Tutoring
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12, Test Prep help for Standardized tests like SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE and AP. You may schedule online tutoring lessons at your personal scheduled times, all with a Money-Back Guarantee. The first one-on-one online tutoring lesson is always FREE, no purchase obligation, no credit card required.
For answers/solutions to any question or to learn concepts, take a FREE CLASS.
No credit card required, no obligation to purchase.
Just book a free class to meet a tutor and get help on any topic you want!
Many acids and bases also act as catalysts, both individually and together. They lend themselves to catalysis on account of their proton-donor and proton-acceptor characteristics. See also: Acid, bases and pH Indicators.
Some examples of acids and bases as catalysts –
- Sulfuric acid is a catalyst used for the decomposition of sucrose into glucose and fructose
- Sulfuric acid is also used as a catalyst in the polymerization of unsaturated hydrocarbons.
- Amorphous silica-alumina, an acid catalyst, is used in the conversion of petroleum hydrocarbons to gasoline.
- Sodium- and potassium-hydroxides are used as catalysts in industrial bio-diesel production
- Base-catalyzed aldol condensation
- Base-catalyzed manufacture of polyurethane foam
Enzymes
Enzymes are naturally occurring proteins that act as catalysts in many biochemical reactions. They are organic catalysts that perform important and critical functions in living beings. The human body has thousands of enzymes enabling respiration, sight, muscle movement, nerve function, digestion and much more.
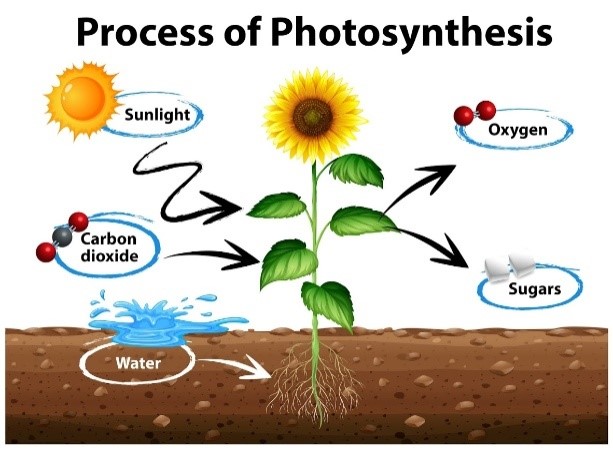
Enzymes at work!
Some enzyme examples –
- Probiotic cultures which ferment milk to make yogurt
- Single-celled micro-organisms called Yeasts produce enzymes which are used in baking breads and pizza dough, in wine-making and many other fermentation processes
- Rennet is used for separating milk into curd and whey and in the production of cheese
- Photosynthesis in plants
- Lipase enzymes help digest fat
- Liver enzymes break down body toxins
- Trypsin breaks proteins into amino acids

Check Point
- Which is not a method to speed up a chemical reaction?
- Heat
- Inert gases
- Use of catalysts
- Concentration of reactants
- Surface area of reactants
- The chemical process of adding a substance to speed up a chemical process is called ______.
- Catalysts are often completely consumed in a chemical reaction. (True/False)
- Many acids and bases act as catalysts. (True/False)
- Naturally occurring proteins that act as catalysts in many biochemical reactions are called ______.
Answer Key
- b) Inert gases
- Catalysis
- False
- True
- Enzymes
Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
Pricing for Online Tutoring
| Tutoring Package | Validity | Grade (1-12), College |
|---|---|---|
| 5 sessions | 1 Month | $139 |
| 1 session | 1 Month | $28 |
| 10 sessions | 3 months | $269 |
| 15 sessions | 3 months | $399 |
| 20 sessions | 4 months | $499 |
| 50 sessions | 6 months | $1189 |
| 100 sessions | 12 months | $2249 |
8th Grade Free Worksheets
- The Universe
- Heredity
- Evolutionary Theory
- Structure of the atom
- Ethical Practices
- Unveiling the mystery behind the physical universe
- Components of the universe
- Celestial phenomena
- The tilt of Earth’s axis
- The causes of high and low tides
- Earth Systems
- Rocks and Fossils
- Weather and Climate
- Basics of chemical reactions
- Types of Chemical reactions – Endothermic, exothermic, oxidation, reduction reactions
- Catalysts and enzymes
- Compounds and mixtures
- Acids, Bases and pH Indicators
Images Credit:
https://lh3.googleusercontent.com/pw/ACtC-3eJ2aA6tkpso0bNudGMGc0EBL4Rh88tBe-p0yZ6BlSZSPQXq32DhYp2kKqLu8vXDp14OSmqb_-dZeXENrbsBUBgXVQaRySi3XpQ_8KrMAe64mzI6_HT2DTVTJtOPKoYAn3x3dbekz28hjVozwlbf_Cq=s867-no
https://lh3.googleusercontent.com/pw/ACtC-3f5cfkZuE-lW1pxC3I67tP6umh5cryIyCafuuswbymSPnQ75VpZkazJG3US1_f7ZlRPCNnqEcWj77np01VyuyFJVxdjDtncQMOm6V44GN44ETbX3PrDMpYkDMUtvTJbbdvOkdVQop1JtwEPbzJUjdU_=w1182-h867-no
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird






