Law of Conservation of Mass Worksheets
Grade 7 Science Worksheets
The Law of Conservation of mass states that “mass can neither be created nor destroyed”.
A system refers to the specific portion of the universe that is under consideration for a particular chemical reaction or physical process. It is a defined area or space where the matter and energy involved in the process are studied. Systems can be classified into two categories – Open System and Closed System.
Example: When some hot drink is poured in a mug, the heat escapes within a few minutes and the mug cools down. So, it is an example of open system.

Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
Open System is a system where a quantity can enter or leave the system to a certain degree.
Example: When a liquid is poured in a thermos flask, there is no exchange of heat or matter. So, it an example of closed or isolated system.
The universe is also an example of isolated system.

Closed Sysytem
Closed System is the opposite of an open system. It is a system where quantity cannot enter or leave the system. Closed system is also called as “isolated system”.
Note: Practically, there is no system which is absolutely closed. Closed systems have limited interaction with the environment.
How is the Law of Conservation of Mass shown by a balanced Equation?
In a chemical reaction, the mass of the reactants (at the start) = mass of the products (at the end)

A better glance at the Law of Conservation of Mass
1. Let‘s say A potter uses 150 grams of clay to make a pot.
When you weigh the pot after its done it is equal to 150 grams.
Weight of the clay used to make the pot = weight of the pot.
Hence the law: Mass can never be created nor is it destroyed.
It just changes its form.

Learn more about Law of Conservation of Mass and other important topics with 7th Grade Science Tutoring at eTutorWorld. Our expert science tutors break down the topics through interactive one-to-one sessions. We also offer the advantage of customized lesson plans, flexible schedules and convenience of learning from home.
eTutorWorld Understands Math Tutoring | Online Math Worksheets are Important Tools
Understanding graphs, charts, and opinion polls in a newspaper, for calculating house and car payments, and for choosing a long-distance telephone service are impossible without strong math skills …and the only way to develop strong math skills is by constant practice.
‘Practice makes a man perfect’ holds true for no other field better than for math. A middle or high school student must set aside a minimum of an hour for math every day. Other than textbooks, worksheets help you revise and understand concepts better.
Our expert tutors prepare online maths worksheets that are age and grade-appropriate. Grade-wise math worksheets for Elementary Math, Arithmetic, Pre-Algebra, Algebra, Geometry, Trigonometry, Statistics, Pre-Calculus and Calculus can be solved to improve math skills, to get ahead or to even catch up.
You may download these FREE online math worksheets in the PDF format, and then print and email us their solutions for a free evaluation and analysis by eTutorworld’smath expert tutors.
You may solve these worksheets by yourself or with your peers while studying together.
The Answer Key at the end of each worksheet allows for a self-evaluation.
Personalized Online Tutoring
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12, Test Prep help for Standardized tests like SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE and AP. You may schedule online tutoring lessons at your personal scheduled times, all with a Money-Back Guarantee. The first one-on-one online tutoring lesson is always FREE, no purchase obligation, no credit card required.
For answers/solutions to any question or to learn concepts, take a FREE CLASS.
No credit card required, no obligation to purchase.
Just book a free class to meet a tutor and get help on any topic you want!
2. Let’s say a cube of ice weighs “x” grams.
When this is weighed after it melts, it weighs the same,
that its weight after melting is “x” grams.
So, weight of the ice cube = weight of the water
Nothing changes with respect to the weight of the ice or water.
It just changes its form.
Mass is neither created not it is destroyed, it’s constant.

3. Experiments involving chemical change also prove the law of conservation of mass.
Weight of (wood + burnt in oxygen) = Weight of the (ashes +carbon dioxide+ water vapor)

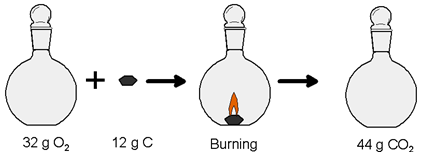
4. How to Prove Law of Conservation of Mass?
Mass of the Reactants = Mass of the Product
Mass of 32 g of oxygen plus 12 g of carbon (before Reaction) = Mass of carbon dioxide (after reaction)
Check Point
A. State True or False
- The law of conservation of mass means, in a closed system, matter cannot be created or destroyed. It changes forms, but is constant.
- Mass of the reactants at the start is always greater than the mass of the product in the end.
- Mass is sometimes viewed as a measure of inertia, the opposition that free bodies offer to forces.
- Matter is always created and destroyed in a closed system.
- The principle of mass conservation, different measurements of the mass of an object taken under various circumstances should always be the same.
B. Explain what does the following mean :
- Mass
- Matter
- Open System
- Closed System
Answer Key
A. True or False:
- True
- False
- True
- False
- True
B. Definitions:
- Mass: Mass is the amount of matter in an object.
- Matter: Matter is anything that has mass and takes up space.
- Open system: a system in which mass or energy can be lost to or gained from its surroundings.
- Closed system: a system in which energy or matter is not exchanged with its surroundings.
What is the Law of Conservation of Mass?
The Law of Conservation of Mass states that in a closed system, the total mass of the substances involved in a chemical reaction remains constant before and after the reaction. This means that matter is neither created nor destroyed during a chemical reaction, only rearranged.
Who discovered the Law of Conservation of Mass?
The Law of Conservation of Mass was proposed by Antoine Lavoisier, a French chemist, in the late 18th century. Lavoisier is often referred to as the “Father of Modern Chemistry” due to his significant contributions to the field.
Is the Law of Conservation of Mass applicable to all types of reactions?
Yes, the Law of Conservation of Mass applies to all types of reactions, including physical changes and chemical reactions. Whether it’s a simple change in state or a complex chemical transformation, the total mass of the substances involved remains constant.
Is the Law of Conservation of Mass applicable to all types of reactions?
Yes, the Law of Conservation of Mass applies to all types of reactions, including physical changes and chemical reactions. Whether it’s a simple change in state or a complex chemical transformation, the total mass of the substances involved remains constant.
Pricing for Online Tutoring
| Tutoring Package | Validity | Grade (1-12), College |
|---|---|---|
| 5 sessions | 1 Month | $139 |
| 1 session | 1 Month | $28 |
| 10 sessions | 3 months | $269 |
| 15 sessions | 3 months | $399 |
| 20 sessions | 4 months | $499 |
| 50 sessions | 6 months | $1189 |
| 100 sessions | 12 months | $2249 |
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
CogAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird






