Law of Conservation of Energy Worksheets
Grade 7 Science Worksheets
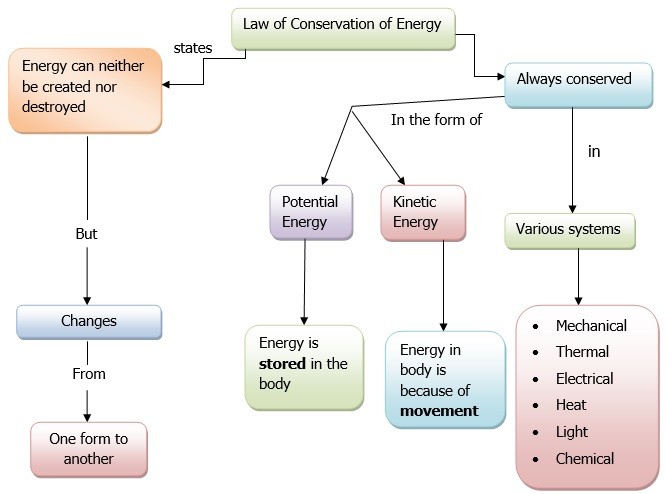
Energy can never be created nor destroyed it just changes form. The law of conservation of energy explains that the total energy in a closed system remains constant it remains to be constant over a period of time.
In a chemical reaction the motion of individual atoms is governed by the law of conservation of energy. In a closed system, a system that is separate from its surroundings, the total energy of the system always remains constant, even if any physical or chemical change happens.
Conservation of energy is the term used to understand change in energy from one form to another.
Credit: http://slideplayer.com/slide/7957227/
Learn more about Law of Conservation of Energy and other important topics with 7th Grade Science Tutoring at eTutorWorld. Our expert science tutors break down the topics through interactive one-to-one sessions. We also offer the advantage of customized lesson plans, flexible schedules and convenience of learning from home.
Schedule a Free session to clear worksheet doubts
No credit card required, no obligation to purchase.
Just schedule a FREE Sessions to meet a tutor and get help on any topic you want!
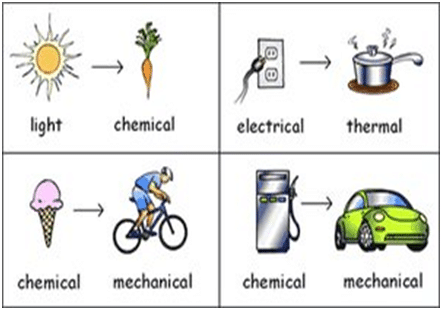
What is an example of the law of conservation of energy?
Burning Candle – Chemical energy—Heat energy/Light energy

Light Bulb – Electrical energy – Heat/light energy

Stove heater —thermal energy—Heat energy/radiant energy

A Flow Chart to Describe Law of Conservation Of Energy
Energy transformation
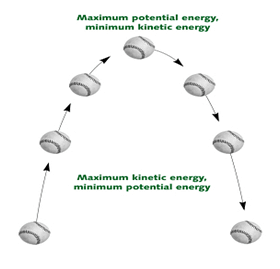
Kinetic energy —- Potential energy —- Kinetic Energy
When a ball is on the ground its velocity is zero.
And its height above the ground is also zero.
Hence the mechanical energy of the whole system is zero.
Now when the ball is thrown up its initial velocity is high and its height above the ground is less.
As the ball is thrown into the sky the speed of the ball falls and also its distance from the earth becomes more that is its height above the ground increases.
But eventually the ball loses its speed and there comes a point where its speed is zero and height is highest.
After this the law of gravity acts on the ball and it starts to fall but now unlike the upward journey the ball gains speed and height becomes lesser.
The ball is constantly changing in the form of energy it contains as it possesses high kinetic energy at the beginning when thrown and finally possesses zero potential energy.
The ball gets kinetically charged as it moves higher later potentially charged.
The ball possesses full potential energy at its highest point, which changes to kinetic energy as it falls to the ground.
As it falls to the ground vice versa happens potential energy converts to kinetic energy.
Kinetic energy is always lost as an object travels upward against the force of gravity and as it loses kinetic energy the object gains potential energy, which in turn converts into kinetic energy as the body falls downward on the Earth.
eTutorWorld Understands Math Tutoring | Online Math Worksheets are Important Tools
Understanding graphs, charts, and opinion polls in a newspaper, for calculating house and car payments, and for choosing a long-distance telephone service are impossible without strong math skills …and the only way to develop strong math skills is by constant practice.
‘Practice makes a man perfect’ holds true for no other field better than for math. A middle or high school student must set aside a minimum of an hour for math every day. Other than textbooks, worksheets help you revise and understand concepts better.
Our expert tutors prepare online maths worksheets that are age and grade-appropriate. Grade-wise math worksheets for Elementary Math, Arithmetic, Pre-Algebra, Algebra, Geometry, Trigonometry, Statistics, Pre-Calculus and Calculus can be solved to improve math skills, to get ahead or to even catch up.
You may download these FREE online math worksheets in the PDF format, and then print and email us their solutions for a free evaluation and analysis by eTutorworld’smath expert tutors.
You may solve these worksheets by yourself or with your peers while studying together.
The Answer Key at the end of each worksheet allows for a self-evaluation.
Personalized Online Tutoring
eTutorWorld offers affordable one-on-one live tutoring over the web for Grades K-12, Test Prep help for Standardized tests like SCAT, CogAT, MAP, SSAT, SAT, ACT, ISEE and AP. You may schedule online tutoring lessons at your personal scheduled times, all with a Money-Back Guarantee. The first one-on-one online tutoring lesson is always FREE, no purchase obligation, no credit card required.
For answers/solutions to any question or to learn concepts, take a FREE CLASS.
No credit card required, no obligation to purchase.
Just book a free class to meet a tutor and get help on any topic you want!
Check Point
A. Fill in the blanks –
- The law of conservation of energy states that the ………… energy of in case of a ………… system remains ………….
- Energy can neither be ………….. nor ………………; rather, it transforms from one form to another.
- Total energy in a closed system remains always ………………
B. State True or False –
- Energy is always created in terms of law of conservation of energy.
- The law of conservation of energy states that the total energy of an isolated system remains constant.
- Law of conservation of energy is a false idea.
- Newton’s laws of motion that the total momentum remains constant in a system completely separated from external factors.
- According to the second law, the momentum of each changes at a rate equal to the force acting on it.
Answer Key
A. Fill in the blanks
- Total, closed , constant
- Created , destroyed
- Constant
B. State true or false
- False
- True
- False
- True
- True
What is the Law of Conservation of Energy?
The Law of Conservation of Energy states that energy cannot be created or destroyed; it can only be transformed from one form to another or transferred between objects or systems.
Why is the Law of Conservation of Energy important?
The Law of Conservation of Energy is a fundamental principle in physics and helps us understand how energy behaves in various systems. It allows us to predict and analyze energy transformations and the behavior of objects and systems.
Does the Law of Conservation of Energy apply to all types of energy?
Yes, the Law of Conservation of Energy applies to all forms of energy, including mechanical, thermal, electrical, chemical, and more. Energy may change from one form to another, but the total amount of energy in a closed system remains constant.
Learn more about Law of Conservation of Energy and other important topics with 7th Grade Science Tutoring at eTutorWorld. Our expert science tutors break down the topics through interactive one-to-one sessions. We also offer the advantage of customized lesson plans, flexible schedules and convenience of learning from home.
Pricing for Online Tutoring
| Tutoring Package | Validity | Grade (1-12), College |
|---|---|---|
| 5 sessions | 1 Month | $139 |
| 1 session | 1 Month | $28 |
| 10 sessions | 3 months | $269 |
| 15 sessions | 3 months | $399 |
| 20 sessions | 4 months | $499 |
| 50 sessions | 6 months | $1189 |
| 100 sessions | 12 months | $2249 |
IN THE NEWS

Our mission is to provide high quality online tutoring services, using state of the art Internet technology, to school students worldwide.
Online test prep and practice
SCAT
CogAT
SSAT
ISEE
PSAT
SAT
ACT
AP Exam
Science Tutoring
Physics Tutoring
Chemistry Tutoring
Biology Tutoring
Math Tutoring
Pre-Algebra Tutoring
Algebra Tutoring
Pre Calculus Tutoring
Calculus Tutoring
Geometry Tutoring
Trigonometry Tutoring
Statistics Tutoring
Quick links
Free Worksheets
Fact sheet
Sales Partner Opportunities
Parents
Passive Fundraising
Virtual Fundraising
Our Expert Tutors
Safe and Secure Tutoring
Interactive Online Tutoring
After School Tutoring
Elementary School Tutoring
Middle School Tutoring
High School Tutoring
Home Work Help
Math Tutors New York City
Press
©2022 eTutorWorld Terms of use Privacy Policy Site by Little Red Bird
©2022 eTutorWorld
Terms of use
Privacy Policy
Site by Little Red Bird